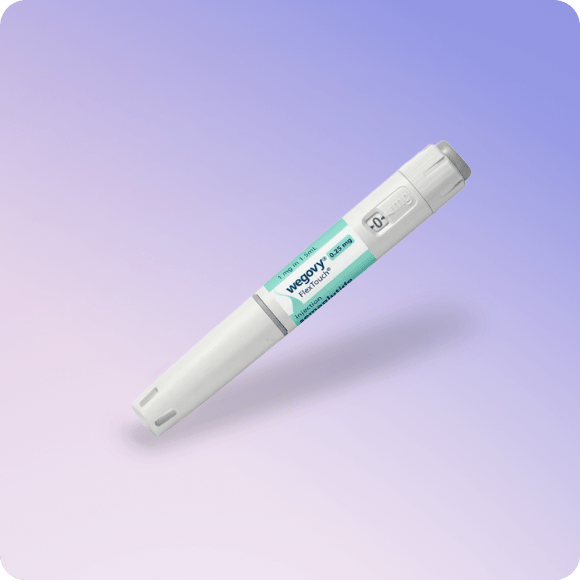


Wegovy (Semaglutide) Weight Loss Injection
Wegovy™ is a once-weekly injectable weight loss aid that is often prescribed when other weight loss efforts have been ineffective. It is only available on prescription.
We provide confidential follow-up and advice to all patients on their weight loss journey.
Priced from £31.25 per week.
- Once Weekly Injection
- Clinically Proven
- Licensed for Weight Loss
- No Hidden Costs
- From £31.25/week
For more information on our delivery times, please click here
Fastest delivery available: Thursday May 2828/05/2026
What is Wegovy (Semaglutide) Weight Loss Injection?
Wegovy is a clinically proven weight loss aid prescribed to help with weight loss when other efforts have been ineffective.
Silence food noise
Targets the reward centre in your brain, so you’ll feel more satisfied after each meal and less likely to experience food focused thoughts.
Feel fuller for longer
Slows down the rate at which your stomach empties, helping to reduce your overall appetite.
Reduce cravings
Keeps your blood sugar level stable after eating, preventing spikes that can lead to increased hunger and cravings.

Overview
Wegovy (semaglutide) is a once-weekly prescription injection that helps adults lose weight when diet and exercise alone are not enough.
It is supplied in a pre-filled pen for easy self-injection under the skin of the stomach, thigh or upper arm.
Wegovy contains semaglutide, a medicine that acts like the natural hormone GLP-1. By supporting the body’s own appetite-control signals, it can help you feel satisfied sooner, reduce food cravings and maintain steadier blood sugar levels.
When combined with a healthy lifestyle, this makes it easier to eat less and achieve gradual, sustained weight loss.
Key Points:
- Prescription only – available after a free online consultation with a UK prescriber
- Once-weekly injection with an easy-to-use pen
- Clinically proven to support significant weight reduction when combined with diet and exercise
- Long-term treatment with gradual dose increases over 16–20 weeks and ongoing maintenance if effective
- Suitable for most adults 18 years and over, subject to a professional assessment
Clinical trials show that people using Wegovy alongside lifestyle changes achieved meaningful, sustained weight loss over time. It’s important to remember that individual results vary.
To find out more about Wegovy, read our guide ‘How Does Wegovy Work for Weight Loss’.
Side Effects
Wegovy™ Side Effects
The most commonly reported side effects in clinical trials were:
- Nausea (reported in 44% of patients)
- Diarrhoea (reported in 30% of patients)
- Vomiting (reported in 24% of patients)
- Constipation (reported in 24% of patients)
Other side effects included abdominal pain, headache, fatigue, and dizziness.
In animal studies, semaglutide was associated with an increased risk of thyroid tumours. This link has not been determined in humans, but Wegovy should not be taken by people with a personal or family history of thyroid tumours or a condition called MEN-2.
Wegovy has been associated with other serious side effects like acute pancreatitis, gallbladder disease, heart rate increase and kidney injury. See the Wegovy information on side effects for further details.
Patient Information Leaflet
Wegovy™ Weight Loss Injection Patient Information Leaflet
For more information about Wegovy™, please take a look at the information leaflet below. It is important you read the Patient Information Leaflet (PIL) before using Wegovy™.
Directions of Use
Wegovy comes in a single-use, prefilled injection pen designed for once-weekly use.
You can inject on any day of the week, at a time that suits you, but try to keep to the same day each week for consistency. It can be taken with or without food.
Using the pen
- Check and prepare – Confirm the pen is in date and at the correct dose. Wash your hands and choose a clean, dry area of skin on the stomach, thigh or upper arm.
- Attach a fresh needle – Screw on a new needle and remove both protective caps.
- Administer the dose – Hold the pen against your skin and press the injection button until you hear or feel the click. Keep the pen in place for the recommended time to ensure the full dose is delivered.
- Dispose needle responsibly – Remove the needle and place it in a sharps container.
Alternate injection sites each week to help prevent irritation.
The pen comes with a full instruction leaflet, which should be read before first use.
To learn more about using Wegovy safely, read our guide ‘Wegovy Interactions and Administration Tips’.
Dosage Schedule
Wegovy treatment begins at a low starting dose and is gradually increased to help your body adjust before reaching the maintenance level.
*INSERT WEGOVY DOSAGE SCHEDULE*
Ingredients
Active ingredient
- Semaglutide (strength varies by pen)
Other ingredients
- Disodium phosphate dihydrate, sodium chloride, hydrochloric acid/sodium hydroxide (pH adjustment), and water for injections.
Check the PIL for the complete ingredient list and speak to a pharmacist if you have any allergies.
Side Effects
Wegovy Side Effects
Like all medicines, Wegovy can cause side effects, although not everyone experiences them.
Common (may affect up to 1 in 10 people):
- Nausea, vomiting or stomach pain
- Diarrhoea or constipation
- Indigestion, bloating or burping
- Decreased appetite
- Tiredness or headache
Less common and very serious:
- Gallstones or gallbladder inflammation
- Pancreatitis (severe abdominal pain, nausea, vomiting)
- Increased heart rate
- Worsening of kidney function (often linked to dehydration from vomiting or diarrhoea)
Seek immediate medical help if you experience severe abdominal pain or symptoms of pancreatitis.
Learn more in our guide “Wegovy Side Effects and How to Avoid Them”.
Warnings
Wegovy may not be suitable for everyone.
Tell your prescriber if you:
- Have a personal or family history of thyroid tumours or a condition called MEN-2
- Have ever had pancreatitis
- Have kidney problems or severe digestive issues
- Are using other GLP-1 medicines such as Saxenda or Ozempic
- Are pregnant, breastfeeding, or planning a pregnancy
Women taking oral contraceptives should use a barrier method (for example, a condom) for 4 weeks after starting Wegovy and after each dose increase.
Stay well hydrated throughout treatment.
Your suitability will be confirmed during the online consultation.
Related guidance:
Alternatives
Wegovy is one of several prescription medicines available in the UK to support weight management.
During your free online consultation, a Cloud Pharmacy prescriber will review your health and help decide which treatment is most suitable for you.
Other options to discuss:
Mounjaro (tirzepatide) – A once-weekly injection that targets two natural hormones to help control appetite and blood sugar.
Saxenda (liraglutide) – A daily injection that helps curb hunger signals.
Orlistat – A capsule taken with meals that reduces the amount of fat your body absorbs.
Mysimba (naltrexone/bupropion) – An oral tablet that works on appetite control centres in the brain.
Xenical (orlistat 120 mg) – The branded version of orlistat with the same active ingredient and effect.
Helpful resources:
- Switching From Mounjaro to Wegovy
- Switching from Saxenda to Wegovy
- Mounjaro vs Wegovy
- Wegovy vs Saxenda
- Wegovy vs Ozempic
- Wegovy Results
- Wegovy Reviews
Reviews
Wegovy (Semaglutide) Weight Loss Injection Reviews
Trusted by 50,000+ patients
Discover more stories

“Your happiness comes from within. If it’s not there, you have to find out how to get it. For me, this was how I found it.”
Read her story

“Minimal side effects, no food noise, and a real sense that this time, something was working.”
Read his story

“I researched it and came across Cloud. It just felt right - It'’s been the best decision for me."
Read her storyHow much weight could you lose with Wegovy in one year
Set your targets with the weight loss calculator. See how much you could lose in a year.
Am I eligible?
Wegovy pricing
If you’re currently using Wegovy (or have recently used it) you can buy the step-up or maintenance doses through Cloud Pharmacy.
| Price | |
| Wegovy 0.25mg | £114.99 |
| Wegovy 0.5mg | £119.99 |
| Wegovy 1mg | £124.99 |
| Wegovy 1.7mg | £149.99 |
| Wegovy 2.4mg | £174.99 |
| Wegovy 7.2mg | £269.99 |
Wegovy (Semaglutide) Weight Loss Injection FAQs
Medically reviewed on August 1st, 2025
What is Wegovy and how does it help with weight loss?
Wegovy contains semaglutide, a GLP-1 hormone analogue that helps reduce appetite, slow stomach emptying and support steady weight loss when used with a healthy diet and exercise.
Who can use Wegovy?
Adults aged 18+ with a BMI of 30 or above, or a BMI of 27+ with a weight-related condition such as high blood pressure, high cholesterol or type 2 diabetes.
How much weight can I lose on Wegovy?
In clinical trials, patients lost an average of 15–17% of their body weight after 68 weeks when combined with diet and exercise.
How is Wegovy taken?
Wegovy is a once-weekly injection administered under the skin of the stomach, thigh or upper arm using a pre-filled pen.
What is the Wegovy dosage schedule?
Doses start at 0.25 mg weekly, increasing every 4 weeks until reaching a maintenance dose of 2.4 mg weekly, if tolerated.
How quickly does Wegovy start working?
Many people notice reduced appetite within a few weeks, with visible weight loss typically appearing after 4–8 weeks.
What are the common side effects of Wegovy?
Nausea, diarrhoea, constipation and vomiting are the most common and often improve over time. Serious but rare effects include pancreatitis and gallbladder disease.
Can I buy Wegovy online in the UK?
Yes. Wegovy is available only on prescription. Cloud Pharmacy offers a free online consultation and next-day UK delivery once approved.
Is Wegovy the same as Ozempic?
Both contain semaglutide, but Ozempic is licensed for diabetes, while Wegovy is licensed for weight management and supplied in different strengths.
How much does Wegovy cost?
Prices vary depending on the dose. Current Wegovy prices are always displayed on this page before you start your consultation.
Can I drink alcohol while taking Wegovy?
Moderate alcohol is not prohibited, but it can worsen stomach side effects. Limit intake and speak to your prescriber if you have concerns.
Can I take Wegovy if I’m pregnant or trying for a baby?
No. Wegovy should be stopped at least two months before a planned pregnancy.
What should I do if I miss a Wegovy dose?
If less than 5 days late, take it as soon as possible and continue your regular schedule. If more than 5 days late, skip the missed dose and resume as planned.
Can I switch from Mounjaro to Wegovy?
Yes, but only under medical supervision. Some patients consider switching from Mounjaro to Wegovy due to recent Mounjaro price increases. Your prescriber will advise on safe timing and dose adjustments.
Find out more by reading our Guide ‘Switching from Mounjaro to Wegovy’.
Do I need a special diet while using Wegovy?
No specific diet is required, but a reduced-calorie meal plan and regular activity will improve results and help maintain weight loss.
A fully qualified team
Our experienced pharmacists are all registered with the General Pharmaceutical Council in the UK.
Wegovy (Semaglutide) Weight Loss Injection guides
Need something else?
We stock over 1102 treatments for 90 conditions
Confirm Wegovy (Semaglutide) Weight Loss Injection treatment selection
| Starting Dose 0.25mg | 1 Month | £114.99 |
| 0.5mg | 1 Month | £119.99 |
| 1.0mg | 1 Month | £124.99 |
| 1.7mg | 1 Month | £149.99 |
| 2.4mg | 1 Month | £174.99 |
| 7.2mg (3x 2.4mg) | 1 Month | £269.99 |
| Starter Park 0.25mg & 0.5mg | 2 Months - 0.25mg & 0.5mg | £229.00 |
| Starter Park 0.25mg & 0.5mg & 1.0mg | 3 Months - 0.25mg & 0.5mg & 1.0mg | £349.00 |














